Ebola vaccines produced lasting antibodies during trial: Studies
Studies in West Africa said that the vaccines developed by Johnson & Johnson and Merck & Co appear to be safe in children and adults.
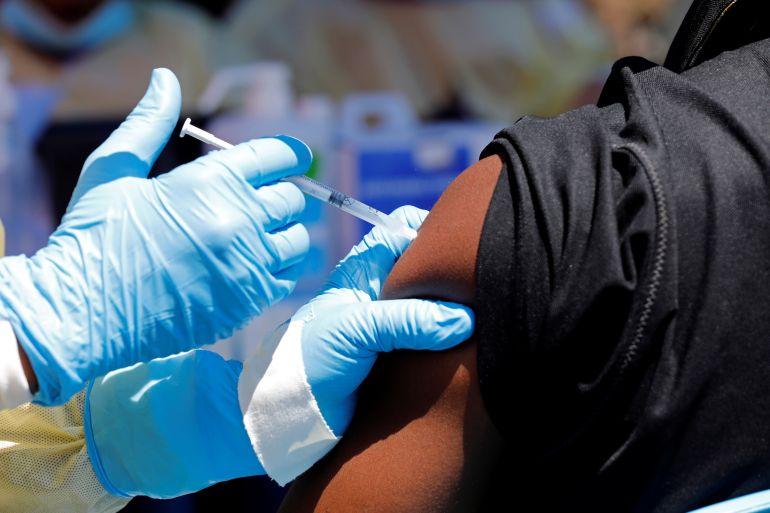
Ebola vaccines developed by Johnson & Johnson and Merck & Co produced virus-fighting antibodies and appear to be safe in children and adults, according to data from two studies conducted in West Africa.
Both companies’ vaccines produced antibodies 14 days after the first of two shots and were detectable at varying levels in both children and adults for one year, data from the studies published on Wednesday showed.
Keep reading
list of 4 itemsUganda declares end of Ebola after 4-month outbreak
Uganda’s Ebola outbreak nearly under control, says Africa CDC
Uganda extends quarantine in Ebola epicentre for 21 days
The results were published in The New England Journal of Medicine.
The vaccines are designed to target the Zaire strain of the virus, not the Sudan strain of Ebola that recently caused an outbreak and at least 56 deaths in Uganda.
One regimen tested a dose of Johnson & Johnson’s vaccine, followed by a booster shot of a vaccine from Danish drugmaker Bavarian Nordic, while another tested two doses of Merck’s vaccine with eight weeks in between. A third option followed the first Merck dose with a placebo.
“I think the study shows that both the vaccines elicit good antibody responses,” said Dr H Clifford Lane, one of the researchers and a clinical director at the United States National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health (NIH).
Only Merck’s shot can potentially be given as a single dose while Johnson & Johnson’s vaccine may need to continue as a two-dose regimen, Lane added.
The NIH researchers noted that they were unable to assess the actual level of protection against the disease from the vaccines as no participants contracted Ebola during the trial, which began enrolment in 2017. But they said the vaccines were found to be safe for children and adults.
“Long-term follow up of the participants in this trial is taking place to determine if and when booster doses might be needed,” said Brian Greenwood, a study co-author from the London School of Hygiene & Tropical Medicine.
A total of 1,400 adults and 1,401 children aged one to 17 years old participated in the trials conducted in collaboration with the Liberia Ministry of Health and with the University Clinical Research Center and the Center for Vaccine Development-Mali.
Merck’s vaccine Ervebo was approved by the European regulator and pre-qualified by the World Health Organization (WHO) in 2019.
Johnson & Johnson’s Zabdeno got European and WHO clearances in 2020 and 2021, respectively.
Bavarian Nordic’s Mvabea, used in Johnson & Johnson’s regimen, also received European approval in 2020 and WHO prequalification in 2021.
The Zaire strain has a mortality rate of 90 percent and is the most lethal of the five Ebola strains. It was the cause of the 2014-2016 Ebola epidemic in West Africa that killed more than 11,000 people.